Clinical update - diagnosis of ARF Fact Checked
DIAGNOSIS OF ARF IN AUSTRALIA
The 2020 Australian guideline for prevention, diagnosis and management of acute rheumatic fever and rheumatic heart disease (3rd edition) contains clinical information based on national and international best practice. The information is supported by a cultural framework to highlight the importance of how care for Aboriginal and Torres Strait Islander people with acute rheumatic fever (ARF) and rheumatic heart disease (RHD) can be both clinically sound, and culturally safe.
In this article, we take a closer look at diagnosis of ARF in the Australian setting.

At risk populations
There is currently no single diagnostic laboratory test for acute rheumatic fever (ARF), therefore, diagnosis is a clinical decision based on presenting symptoms and signs, as well as personal history of ARF and/or rheumatic heart disease (RHD) and some laboratory parameters. Diagnosis of ARF also varies according to level of risk. For example, in a region with a high incidence of ARF, a person with fever and arthritis is more likely to have ARF than someone in a low-incidence region. In Australia, ARF is more common in Aboriginal and Torres Strait Islander peoples.
Classifying someone into a risk group for ARF is important, because there are implications for the diagnostic criteria for individuals at high and low risk. People considered to be at high risk of ARF are more likely to live in an area with high rates of ARF, are more likely to live in rural, remote and/or crowded settings, and/or are more likely to have a history of ARF or RHD (2020 guideline Table 6.1 page 73).1

Australians who do not fit into the above categories are considered to be at low risk for ARF.
Criteria for ARF diagnosis
The Jones criteria for the diagnosis of ARF were developed in the USA and introduced in 1944.2 Major manifestations including arthritis, carditis Sydenham chorea, erythema marginatum and subcutaneous nodules are more closely associated with ARF, and minor manifestations such as fever and raised ESR or CRP are indicators of generalised inflammation which are used to support the diagnosis. While the Jones criteria have been periodically modified and updated since 1944, mainly to cater to the declining incidence in north America, they are not adequately sensitive to identify ARF in persisting high-incidence populations, such as in Australia. For example, symptoms and signs of ARF in Aboriginal and Torres Strait Islander people are not necessarily consistent with the 1992 version of the Jones criteria.3,4,5 To help overcome this, diagnostic criteria for high and low risk groups have been separated over recent years,6 and this separation initially developed in Australia has been adopted globally.
Several changes were introduced in the 2020 Australian guideline (Table 6.2, page 74) to align with the 2015 American Health Association revised Jones criteria7 and facilitate a global standard for ARF diagnosis.8 The recent changes relate specifically to low-risk groups and include:
- subclinical carditis as a major diagnostic criterion.
- erythrocyte sedimentation rate (ESR) as a minor criterion is now ≥60 mm/h for low-risk groups.
- fever as a minor criterion is now ≥38.5°C for low risk-groups.
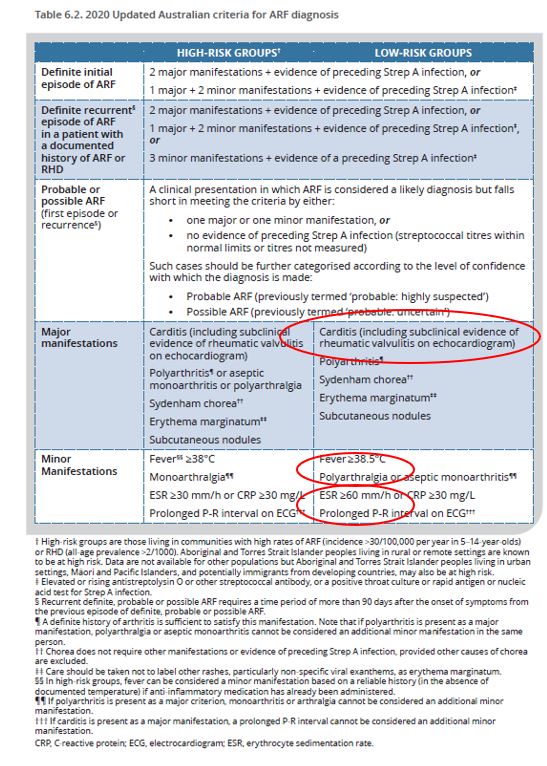
For more information about the major and minor criteria of ARF, see Table 6.6 (page 86) and Table 6.7 (page 89), respectively.
For everyone, a definite recurrent episode of ARF with a documented history of ARF or RHD now requires 2 major, or 1 major and 2 minor (not 1 minor), or 3 minor criteria, plus evidence of preceding Strep A infection.
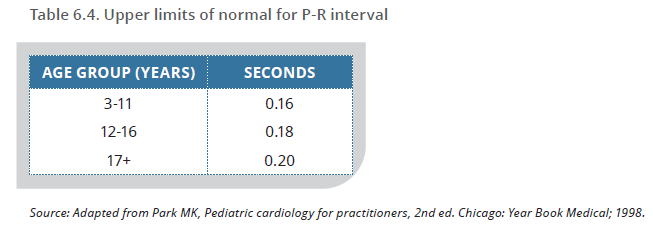
The following table from the 2020 guideline (page 75) 9 provides guidance for interpreting P-R interval on electrocardiogram.
Difficulties with ARF diagnosis
ARF can be difficult to diagnose. Symptoms may be mild or atypical, or there may be a delay in presentation to the health service, all of which make investigation and interpretation difficult.10 Getting the diagnosis right is important, because failure to diagnose ARF is a missed opportunity for secondary prophylaxis, increasing the risk of recurrent ARF and RHD, and over-diagnosis results in unnecessary treatment for the individual and unnecessary use of health system resources.
Medical and nursing staff working in areas with high rates of ARF should have a heightened level of suspicion and be familiar with recommended management of people suspected to have ARF. Communities with high rates of disease should be supported to increase their understanding of the risk factors and symptoms of ARF, so that they are diagnosed quickly and managed appropriately.
| All people suspected to have ARF should be admitted to hospital within 24 hours where possible, for timely blood testing, echocardiography and specialist review, monitoring of fever and joint symptoms, and disease education. Family members should be included wherever possible. |

RHDAustralia has developed an ARF diagnosis app to guide clinicians who suspect ARF. The app works through levels of risk including history of ARF and RHD and presenting symptoms and signs and provides a recommendation for next steps. Despite the availability of clear diagnostic criteria for ARF, there are several considerations when using the formula:
- If polyarthritis is present as a major criterion, monoarthritis or arthralgia cannot be considered an additional minor manifestation.
- If carditis is present as a major manifestation, a prolonged P-R interval cannot be considered an additional minor manifestation.
- Provided other causes of chorea are excluded, Sydenham chorea alone (i.e., without other symptoms or evidence of recent Strep A infection) can be used to confirm ARF.
For some people, ARF cannot be confirmed or refuted and to avoid or minimise the impact of potential RHD, these people should be managed according to the protocol for probable or possible ARF, based on the diagnostic criteria.11
- Probable ARF - an acute presentation which does not fulfil Jones diagnostic criteria for ARF, missing one major or one minor criterion or lacking evidence of preceding streptococcal infection, but where ARF is still considered the most likely diagnosis.
- Possible ARF – an acute presentation which does not fulfil Jones diagnostic criteria for ARF, missing one major or one minor criterion or lacking evidence of preceding streptococcal infection, and where ARF is considered uncertain but cannot be ruled out.
| Echocardiogram is mandatory for all people with possible or confirmed ARF, even if the criteria for notification of ARF have been met. Echocardiogram can identify subtle carditis, assess the severity of valvulitis and cardiac function, establish the presence of pre-existing, undiagnosed RHD, and serve as a baseline for future monitoring of disease progression (given the high rate of disease progression to RHD12) and/or recurrent ARF. |
Evidence of Streptococcal A infection
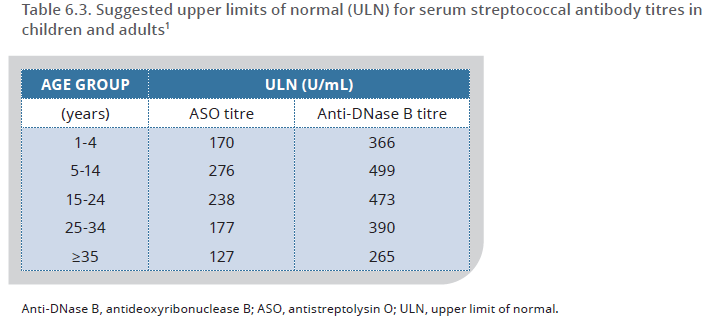
Evidence of a recent Strep A infection completes the recipe for ARF diagnosis, and swabbing the throat is a relatively easy way to collect a sample. However, isolating Strep A from a throat swab is not reliable. For example, in New Zealand, 12.6% of school-aged children with sore throat returned Strep A,13 whereas in Uganda, Strep A was confirmed in 41.8% of children with sore throat.14 Therefore, the most reliable tests to identify Strep A are the ASO and the anti-DNase B titres, which both require a blood sample.
The serum ASO titre usually rises within one to two weeks after infection, and peaks at about three to six weeks, while the serum anti-DNase B titre can take up to six to eight weeks to peak.15 The rate of decline of these antibodies varies enormously, with the ASO titre starting to fall at six to eight weeks, and the anti-DNase B titre falling around three months after infection.16 If there is no re-infection with Strep A, the ASO titre usually approaches pre-infection levels after 6–12 months, whereas the anti-DNase B titre tends to remain elevated for longer - sometimes indefinitely - during childhood, especially in communities with high rates of Strep A skin infections.17
Streptococcal titres vary according to several factors, including age (see Table 6.3, page 75) below.
Streptococcus A rapid diagnostics
Strep A rapid tests include rapid antigen detection tests (RADT) and recently developed molecular tests for detecting Strep A DNA. Current RADT are not as accurate as culture for detecting Strep A,18 with limited use in children and in public health interventions for control of ARF and RHD. Where RADT are used – not routinely in Australia - clinical practice guidelines recommend a negative test is followed by a backup throat swab culture.19 A future potential role for the more sensitive Strep A molecular detection tests is currently being studied.
- 1. RHDAustralia (ARF/RHD writing group). The 2020 Australian guideline for prevention, diagnosis and management of acute rheumatic fever and rheumatic heart disease (3rd edition); 2020. View Source
- 2. Jones T. Diagnosis of rheumatic fever. Journal of the American Medical Association 1944; 126: 481-4
- 3. Stewart T, McDonald R, Currie B. Use of the Jones criteria in the diagnosis of acute rheumatic fever in an Australian rural setting. Australian and New Zealand Journal of Public Health 2005; 29(6): 526-9
- 4. Ralph A, Jacups S, McGough K, et al. The challenge of acute rheumatic fever diagnosis in a high-incidence population: a prospective study and proposed guidelines for diagnosis in Australia’s Northern Territory. Heart Lung and Circulation 2006; 15(2): 113-8
- 5. Noonan S, Zurynski YA, Currie BJ, et al. A national prospective surveillance study of acute rheumatic fever in Australian children. The Pediatric Infectious Disease Journal 2013; 32(1): e26-32
- 6. RHDAustralia (ARF/RHD writing group), National Heart Foundation of Australia and the Cardiac Society of Australia and New Zealand. Australian guideline for prevention, diagnosis and management of acute rheumatic fever and rheumatic heart disease (2nd edition). 2012
- 7. Gewitz MH, Baltimore RS, Tani LY, et al. Revision of the Jones Criteria for the diagnosis of acute rheumatic fever in the era of Doppler echocardiography: a scientific statement from the American Heart Association. Circulation 2015; 131(20): 1806-18
- 8. RHDAustralia (ARF/RHD writing group). The 2020 Australian guideline for prevention, diagnosis and management of acute rheumatic fever and rheumatic heart disease (3rd edition); 2020. View Source
- 9. RHDAustralia (ARF/RHD writing group). The 2020 Australian guideline for prevention, diagnosis and management of acute rheumatic fever and rheumatic heart disease (3rd edition); 2020. View Source
- 10. Noonan S, Zurynski YA, Currie BJ, et al. A national prospective surveillance study of acute rheumatic fever in Australian children. The Pediatric Infectious Disease Journal 2013; 32(1): e26-32
- 11. RHDAustralia (ARF/RHD writing group). The 2020 Australian guideline for prevention, diagnosis and management of acute rheumatic fever and rheumatic heart disease (3rd edition); 2020. View Source
- 12. He VYF, Condon JR, Ralph AP, et al. Long-term outcomes from acute rheumatic fever and rheumatic heart disease: A data-linkage and survival analysis approach. Circulation 2016; 134:222-32
- 13. Oliver J, Upton A, Jack S, et al. Distribution of Streptococcal Pharyngitis and Acute Rheumatic Fever, Auckland, New Zealand, 2010–2016. Emerging Infections Diseases 2020; 26(6):1113–1121.
- 14. DeWyer A, Scheel A, Webel AR, et al. Prevalence of group A β-hemolytic streptococcal throat carriage and prospective pilot surveillance of streptococcal sore throat in Ugandan school children. International Journal of Infectious Diseases, 2020; 93:245-251.
- 15. Kaplan E, Ferrieri P, Wannamaker LW. Comparison of the antibody response to streptococcal cellular and extracellular antigens in acute pharyngitis. Journal of Paediatrics, 1974. 84(1): 21-8.
- 16. McCarty M. The antibody response to streptococcal infections, in Streptococcal infections, Columbia University Press: New York. 1954 p. 130-42.
- 17. Stollerman G, Lewis AJ, Schultz I, et al. Relationship of immune response to group A streptococci to the course of acute, chronic and recurrent rheumatic fever. American Journal of Medicine, 1956. 20(2): 163-9.
- 18. Lean WL, Arnup S, Danchin M, Steer AC. Rapid diagnostic tests for group A streptococcal pharyngitis: a meta-analysis. Pediatrics 2014; 134(4): 771-81
- 19. Shulman ST, Bisno AL, Clegg HW, et al. Clinical Practice Guideline for the Diagnosis and Management of Group A Streptococcal Pharyngitis: 2012 Update by the Infectious Diseases Society of America. Clinical Infectious Diseases 2012; 55: e86-e102.
